
No2 Lewis Structure
A step-by-step explanation of how to draw the NO2 - Lewis Dot Structure (Nitrite ion). For the NO2 - structure use the periodic table to find the total number of valence electrons f.
Chemical structure nitrogen dioxide no2 Stock Vector Images Alamy
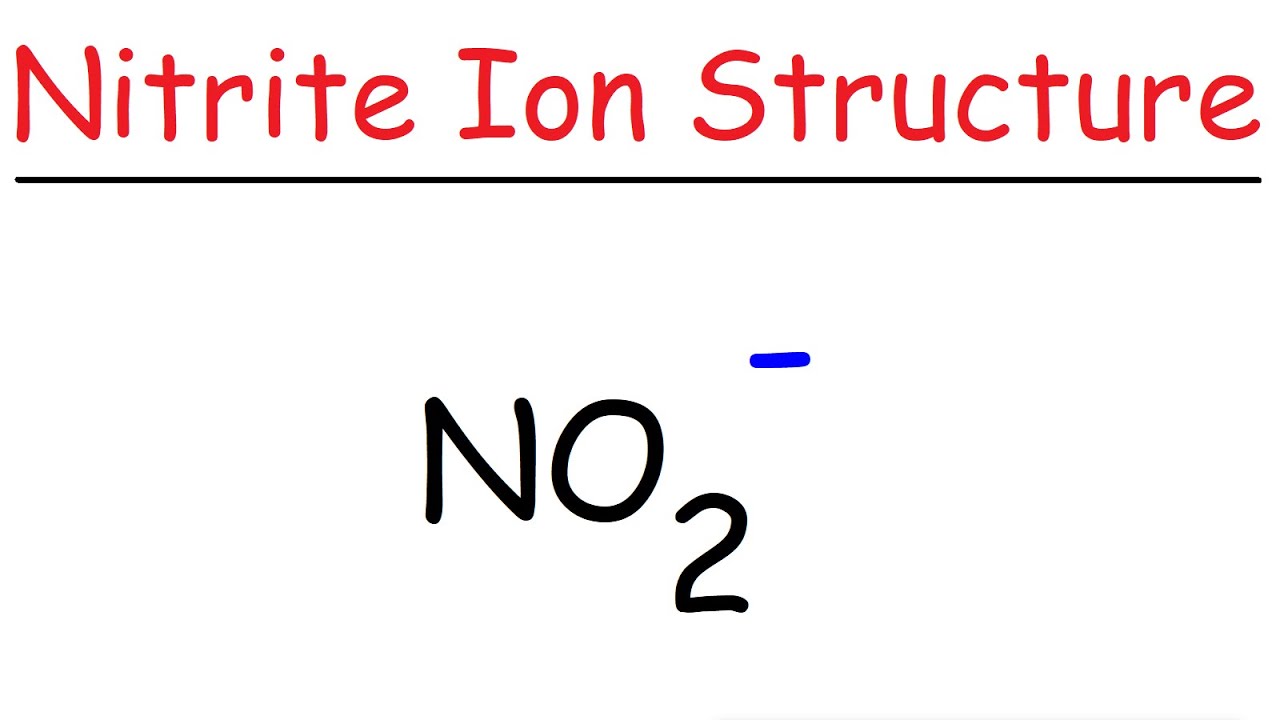
NO 2 structure. The bond length between the nitrogen atom and the oxygen atom is 119.7 pm. That means both bonds exist between oxygen and nitrogen are same. Due to exist of unpaired electron, NO 2 is a free radical. There is a unpaired electron on nitrogen atom in NO2 lewis structure.

NO2 Molecular Geometry / Shape and Bond Angles YouTube
Welcome to Warren Institute! In this article, we will dive into the fascinating world of Chemistry and explore the NO2- Lewis Structure. As aspiring
NO2 Lewis Structure Nitrite Ion YouTube
This chemistry video tutorial explains how to draw the lewis structure of NO2-, the Nitrite ion.Chemistry - Basic Introduction: https://ww.

Nitrogen dioxide, NO2, molecule model and chemical formula ⬇ Vector
NO2 - Molecular Geometry / Shape and Bond Angles Wayne Breslyn 727K subscribers Join Subscribe Subscribed 556 Share 149K views 4 years ago A quick explanation of the molecular geometry of NO2 -.
NO2+ Lewis Structure Nitronium Ion YouTube
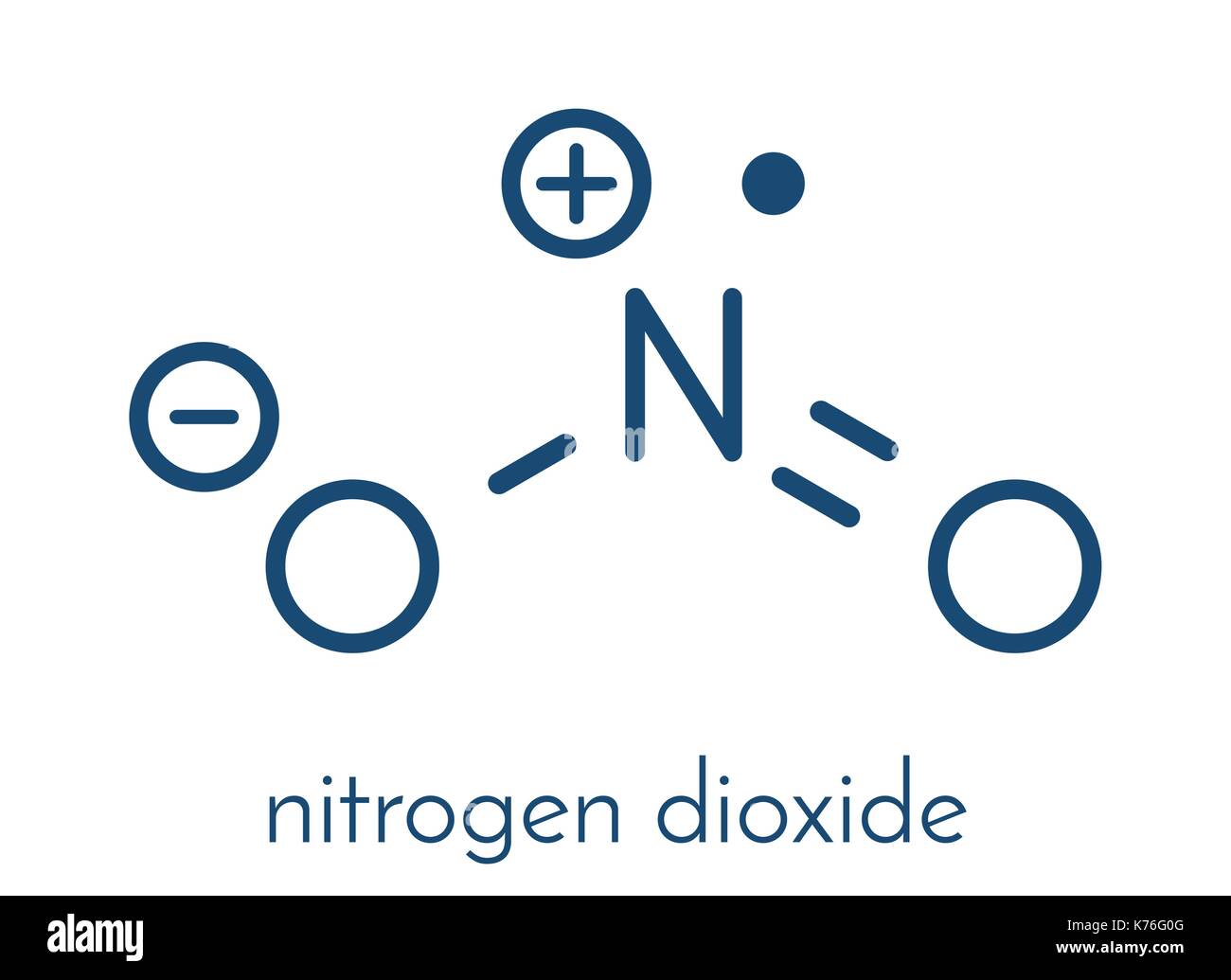
The Lewis structure of NO2, which consists of one nitrogen atom bonded to two oxygen atoms with a double bond, influences its chemical properties. This structure implies that NO2 has a linear geometry and a formal charge of +1 on the nitrogen atom and -1 on each oxygen atom.
No2 lewis structure
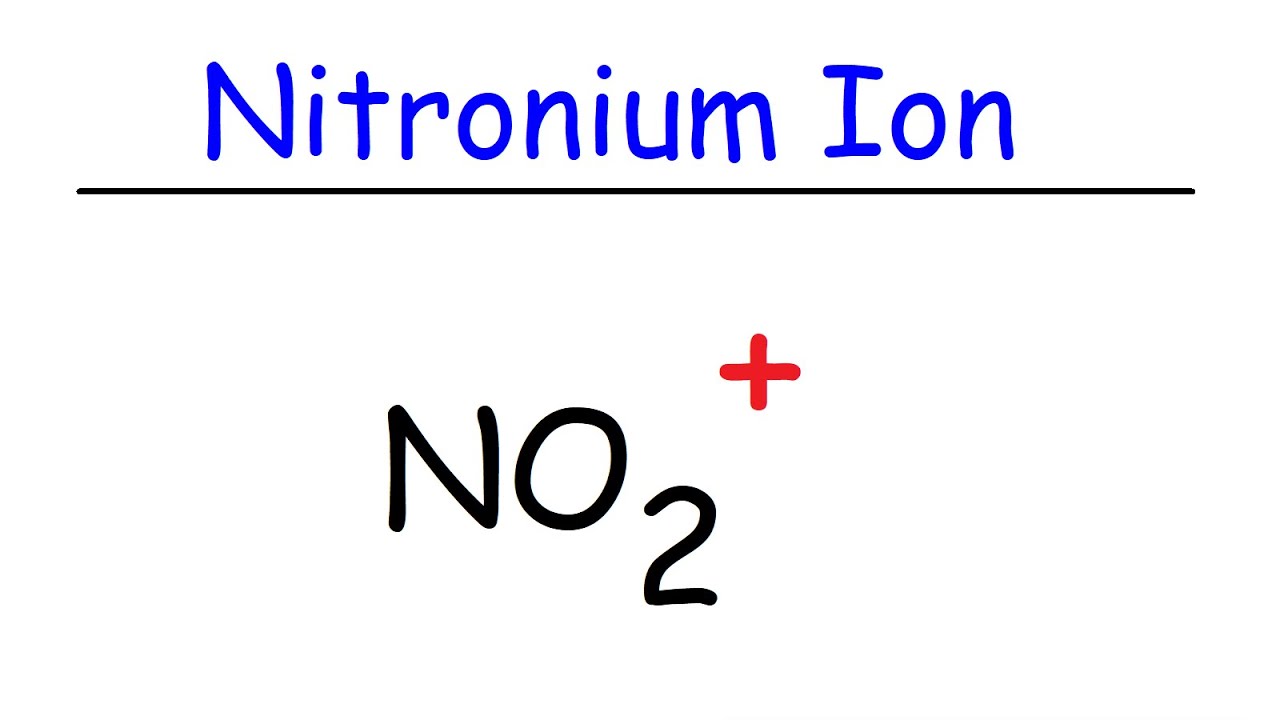
1.1K 208K views 6 years ago A step-by-step explanation of how to draw the NO+ Lewis Dot Structure (Nitronium ion). For the NO+ structure use the periodic table to find the total number of.

How To Draw The Lewis Structure of NO2+ (Nitronium Ion) YouTube
Steps Sketch the structure Location of nitrogen and oxygen on the periodic table To sketch the NO 2 Lewis structure, the first step is to determine the total number of valence electrons in the molecule. This can be calculated by multiplying the valence electrons of each atom.

Lewis Structure of NO2(1), the nitrite ion. YouTube
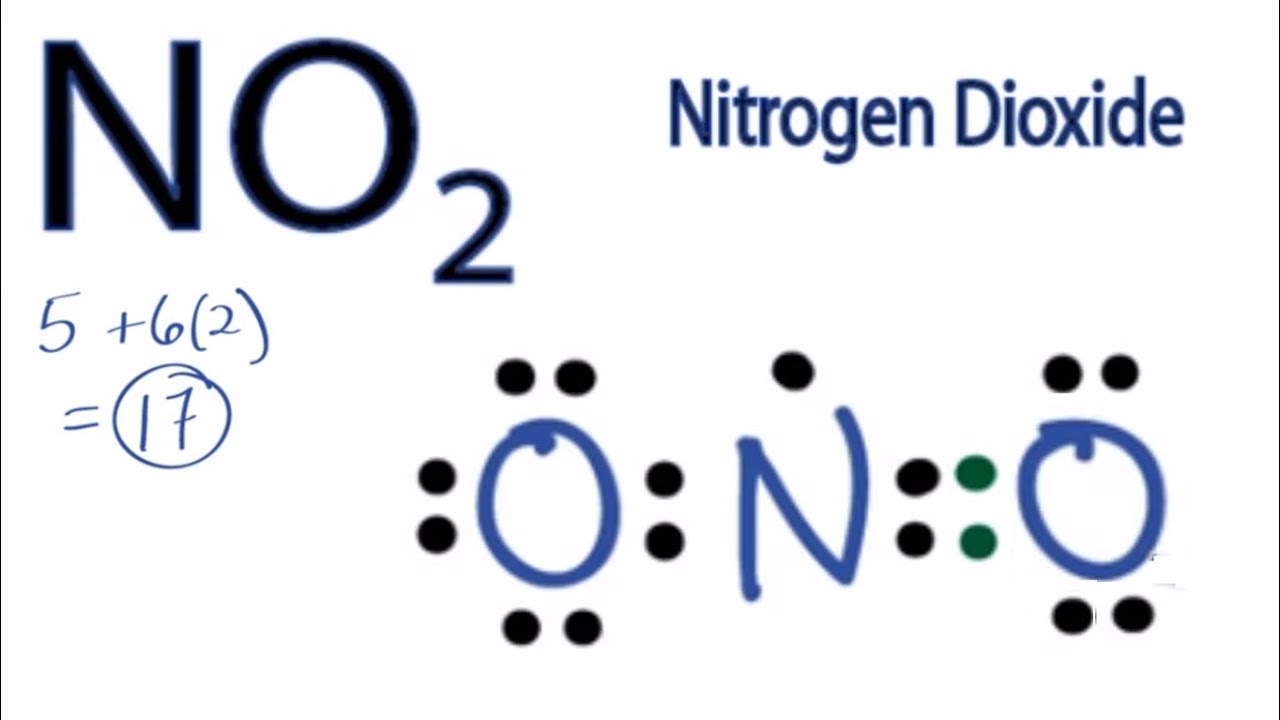
Let us talk about drawing the Lewis Structure for Nitrogen Dioxide ( NO2 ). Lewis Structure of NO2 A molecule of nitrogen dioxide consists of one nitrogen atom and two atoms of oxygen. Let us look at the periodic table. Nitrogen belongs to group 15 ( or group 5) and has an atomic number of 7, therefore has a valency of 5.

How to draw NO2+ Lewis Structure? Science Education and Tutorials
Nitrogen dioxide is a chemical compound with the formula NO2. One of several nitrogen oxides, nitrogen dioxide is a reddish-brown gas. It is a paramagnetic, bent molecule with C 2v point group symmetry.
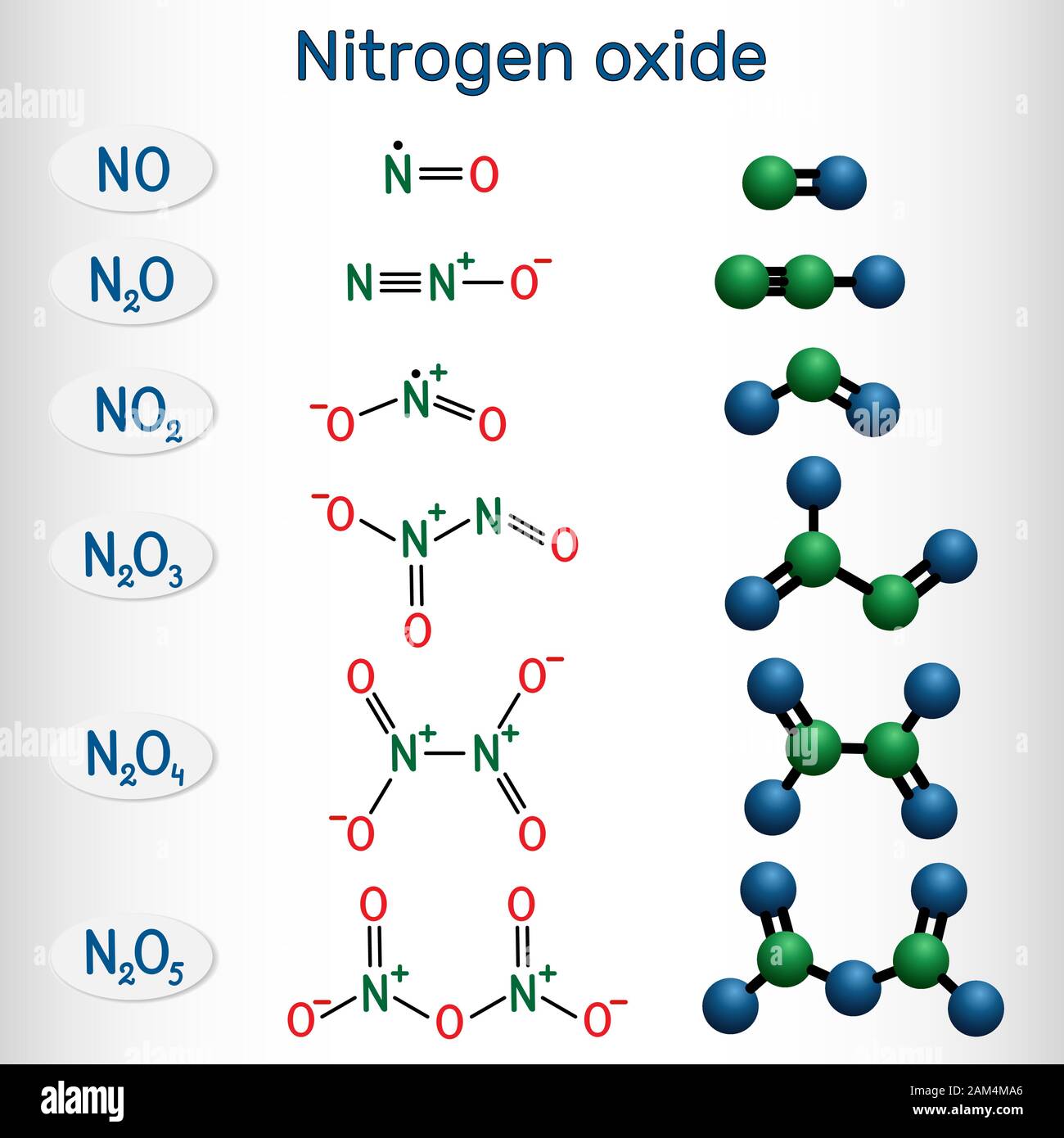
Chemical formulas and molecule model of nitrogen oxide nitric oxide NO
This video explains how to draw the lewis structure of the nitronium ion NO2+.Chemistry - Basic Introduction: https://www.youtube.com/watc.
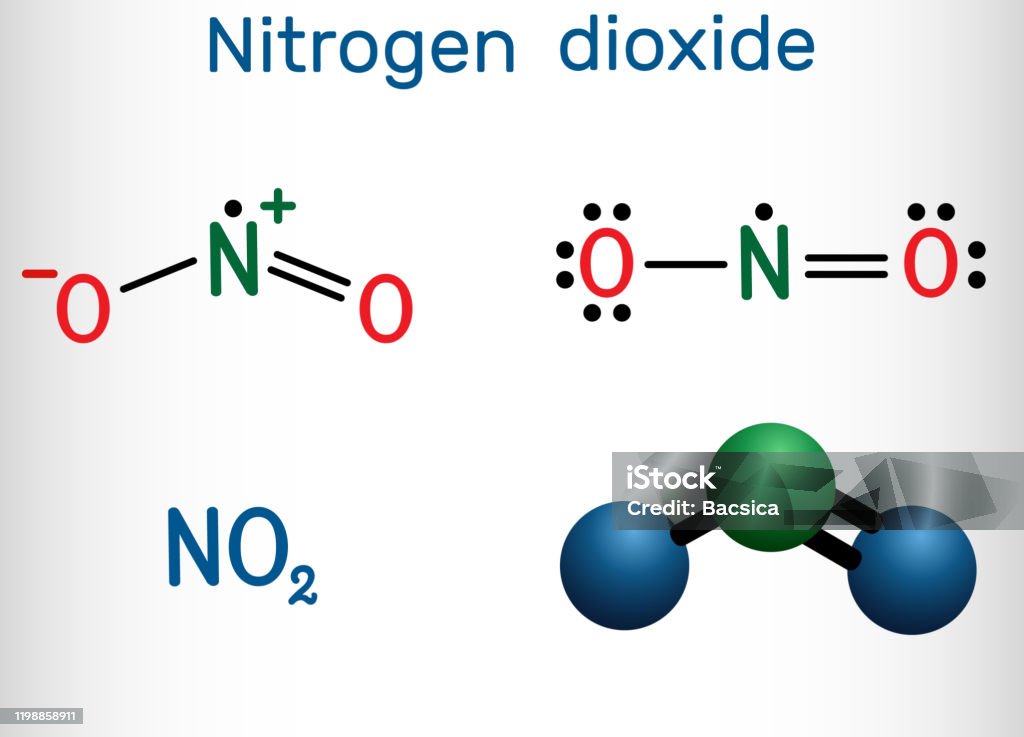
Vetores de Dióxido De Nitrogênio Molécula No2 Fórmula Química
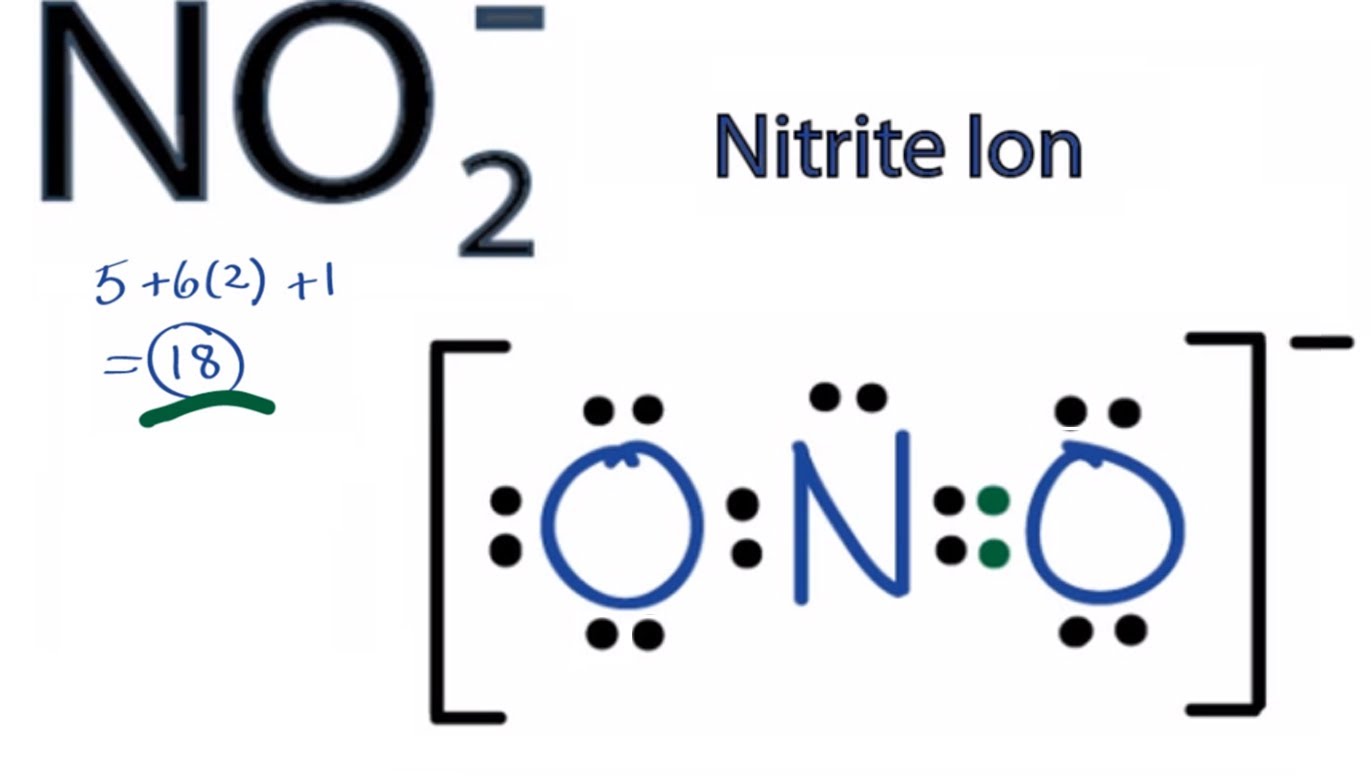
Lewis dot structure: A Lewis Structure, also known as an Electron Dot Structure, is a simplified representation of a molecule's valence shell electrons. It describes the arrangement of valence electrons around particular atoms in a molecule. Electronic configuration of nitrogen = 1 s 2 2 s 2 2 p 3 and electronic configuration of oxygen = 1 s 2.

NO2 Lewis Structure, Molecular Geometry, Hybridization, Polarity, and
The Lewis structure of a NO2- consists of a nitrogen (N) atom and two oxygen (O) atoms. The nitrogen (N) atom is present at the center of the molecular ion, while the two O-atoms occupy terminal positions. Meanwhile, there is a lone pair of electrons on the central N-atom.
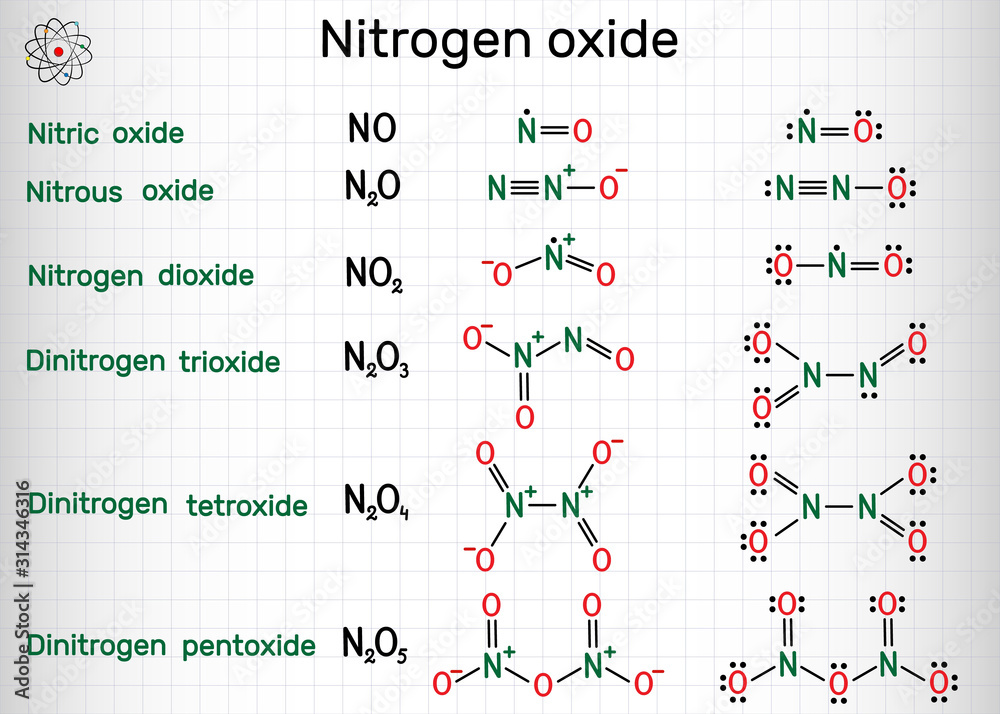
Chemical formulas of nitrogen oxide nitric oxide NO, nitrogen dioxide
NO 2 molecule: The Lewis structure of NO 2 molecule is shown below. Figure 1.2h NO2 molecule Lewis structure. For above molecules, they all contain unpaired (single) electrons. The neutral species that contain an unpaired electron is called radical (or free radical). When the carbon atom of a alkyl group has an unpaired electron, the species is.
Drawing Lewis Structures Chemistry Socratic
Subscribed 2.4K 478K views 10 years ago A step-by-step explanation of how to draw the NO2 Lewis Structure (Nitrogen Dioxide). The NO2 Lewis structure has a total of 17 valence electrons..

Estructura de Lewis NO2, Ejercicios Resueltos » Quimica Online
In the Lewis structure for NO2 the Nitrogen atom is the least electronegative atom and goes at the center of the structure. For the NO2 Lewis structure, calculate the total number of valence electrons for the NO2 molecule. After determining how many valence electrons there are in NO2, place them around the central atom to complete the octets.